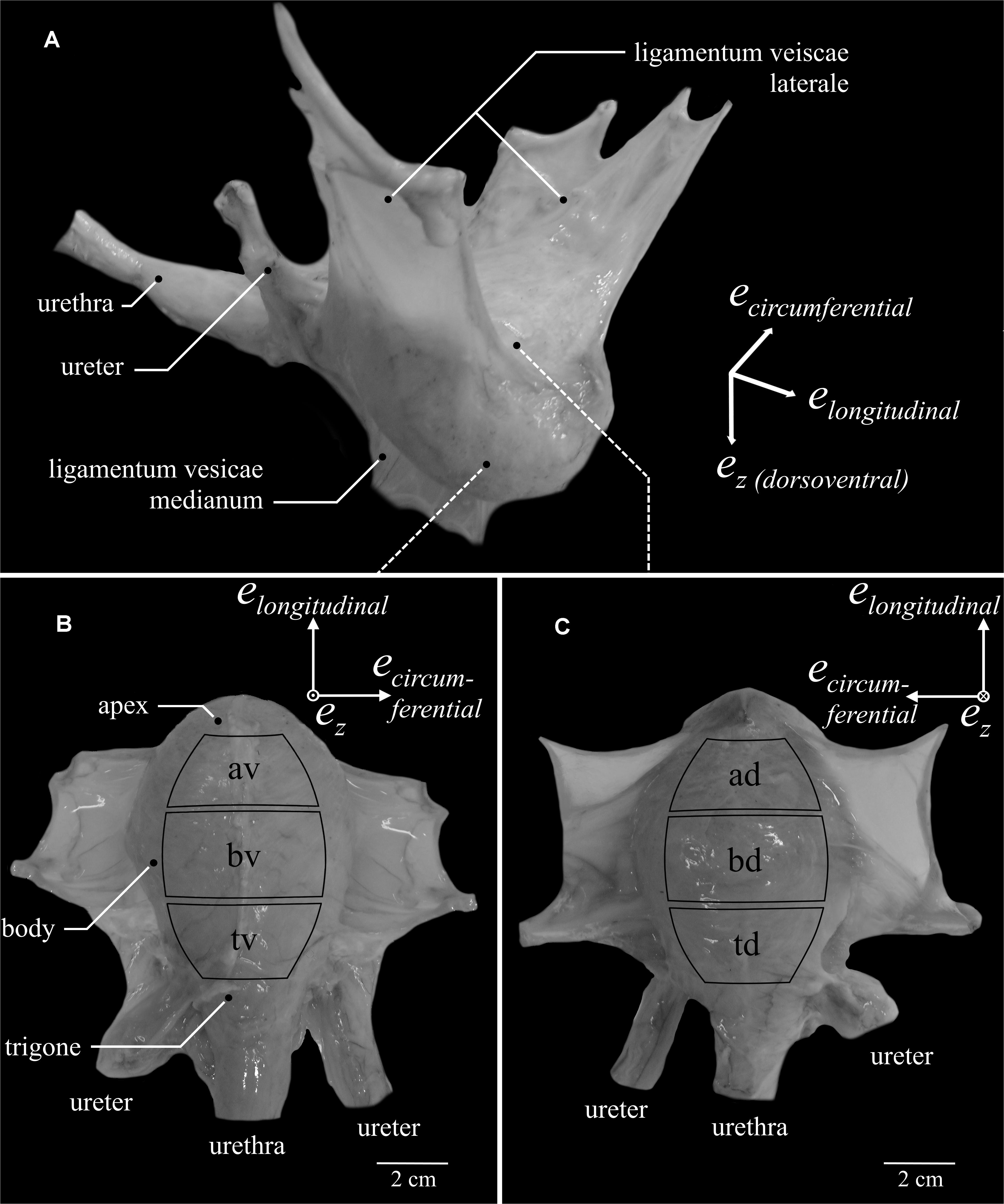
Frontiers | Locational and Directional Dependencies of Smooth Muscle Properties in Pig Urinary Bladder

Estimating the effect of increasing dispensing intervals on retention in care for people with HIV in Haiti - eClinicalMedicine

Zincate‐Mediated Remote Functionalisation of p‐Iodobenzyl Derivatives Through Metallotropy in 2‐Methyltetrahydrofuran as Key Solvent - Pierret - 2022 - Advanced Synthesis & Catalysis - Wiley Online Library

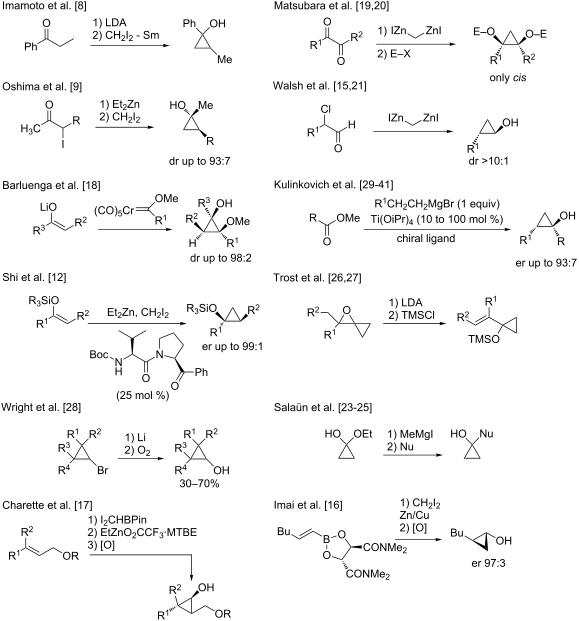
Streamlined Catalytic Enantioselective Synthesis of α-Substituted β,γ-Unsaturated Ketones and Either of the Corresponding Tertiary Homoallylic Alcohol Diastereomers | Journal of the American Chemical Society
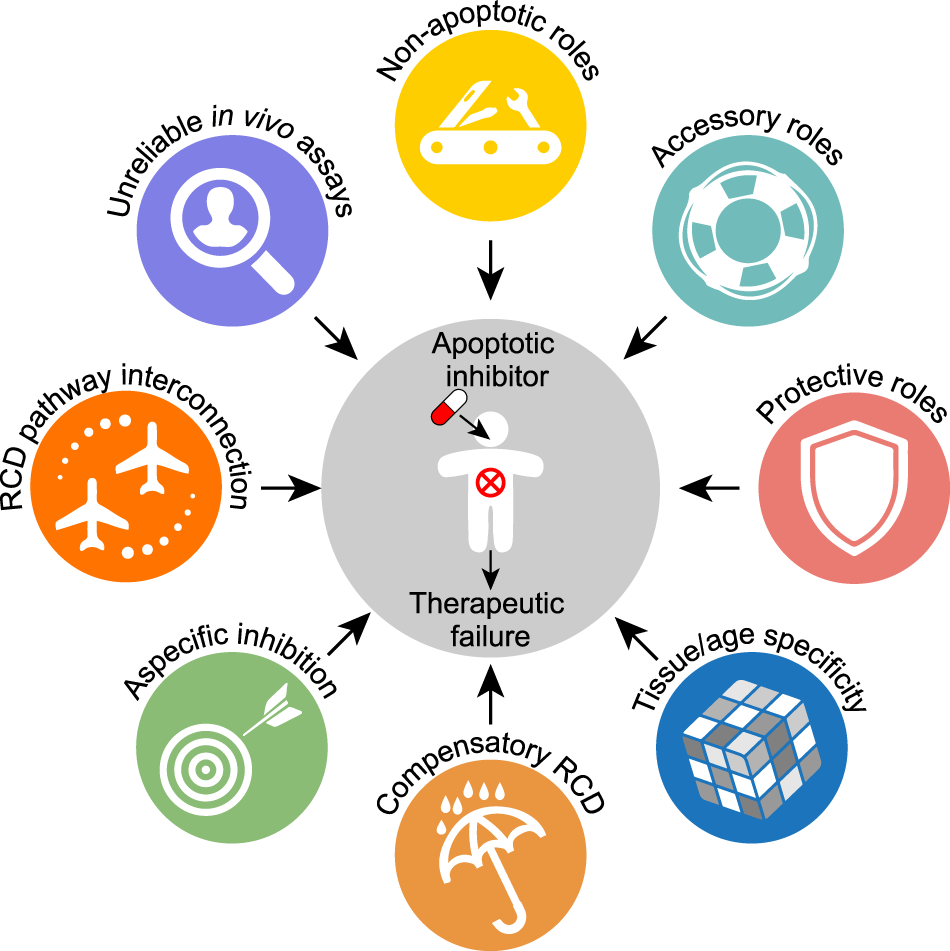
Apoptotic cell death in disease—Current understanding of the NCCD 2023 | Cell Death & Differentiation

PDF) ChemInform Abstract: Addition Reactions of Chloro- or Iodomethyllithium to Imines. Synthesis of Enantiopure Aziridines and β-Chloroamines.

Asymmetric Synthesis of Axially Chiral C−N Atropisomers - Rodríguez‐Salamanca - 2022 - Chemistry – A European Journal - Wiley Online Library

A Reverse Approach to the Total Synthesis of Halichondrin B | Journal of the American Chemical Society

Pharmaceuticals | Free Full-Text | Glycoconjugates of Mucochloric Acid—Synthesis and Biological Activity